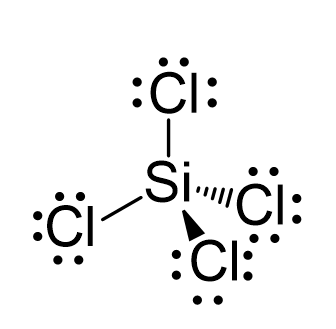
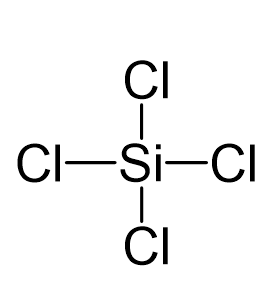
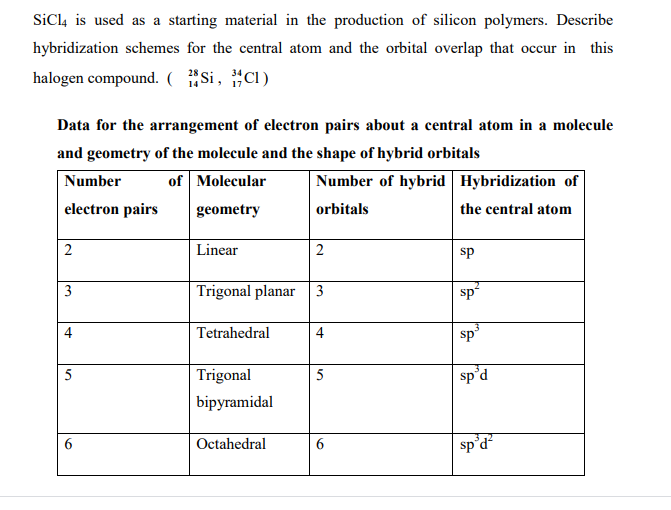
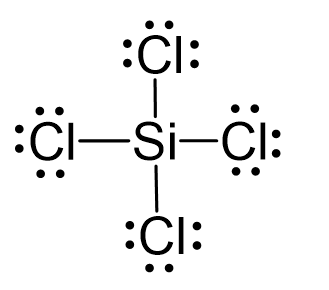
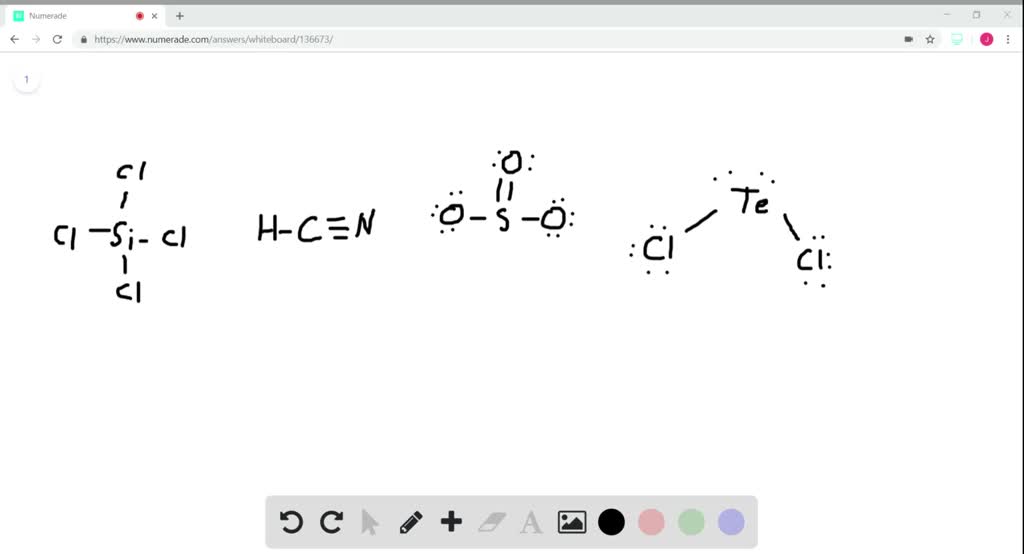
SiCl4 lewis structure, molecular geometry, hybridization, polar or nonpolar | Molecular geometry, Molecular shapes, Molecular
Hybridization is a phenomenon that takes place in an atom before chemical bonding. How is hybridization responsible for the observed structure of SiCl4? - Quora

SOLVED: 4a) Using Valence Bond Theory, show the hybridization and bonding scheme for silicon tetrachloride (SiCl4): (a) write the atomic orbital diagram for the central atom, (b) circle the atomic orbitals that
In case of hybridization in chemical bonding is only the central atom hybridized or atoms also except central atoms hybridize to form bonds? What is the exact concept of hybridization? - Quora
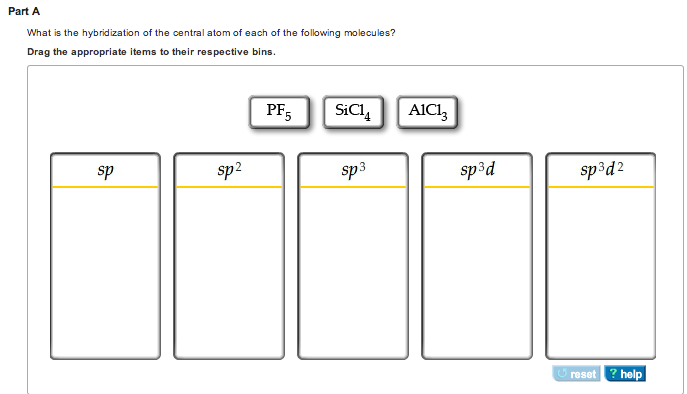
What is the hybridization of the central atom of each of the following molecules? - Home Work Help - Learn CBSE Forum
We all know that CCl4 and SiCl4 both compound remains in Tetrahedral structure , with a hybridization of sp³ in the central atom. Now , Si is a much larger atom than