Improvement of the Stability of IO3–-, SeO32–-, and SeO42–-Coprecipitated Barite after Treatment with Phosphate | Environmental Science & Technology
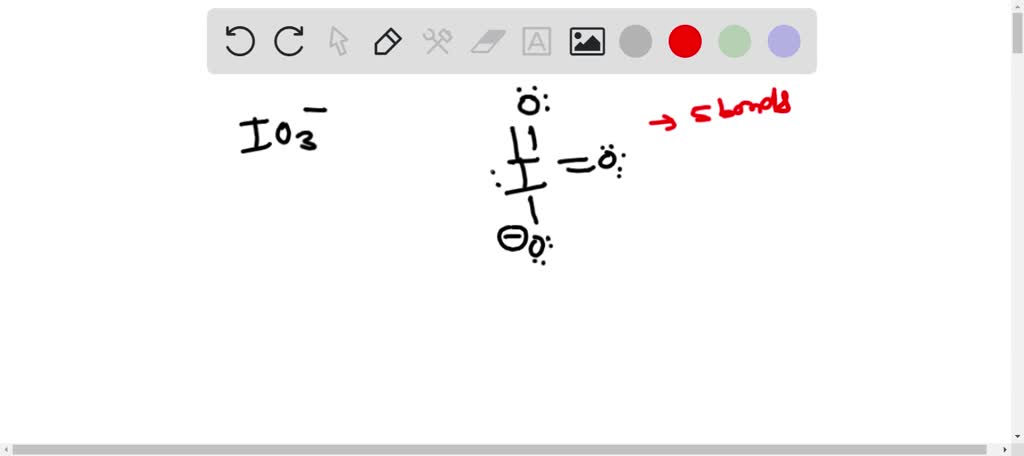
SOLVED: Draw the Lewis structure for the IO3- ion before answering the following questions. Iodine is the central atom and all atoms obey the octet rule. How many bonds does the Iodine
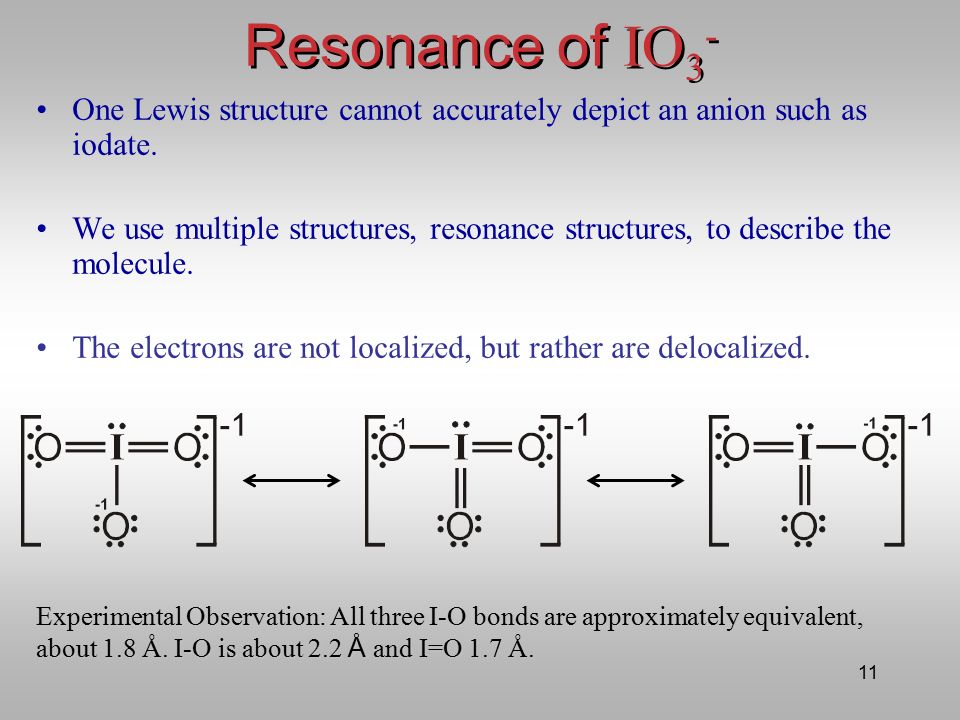
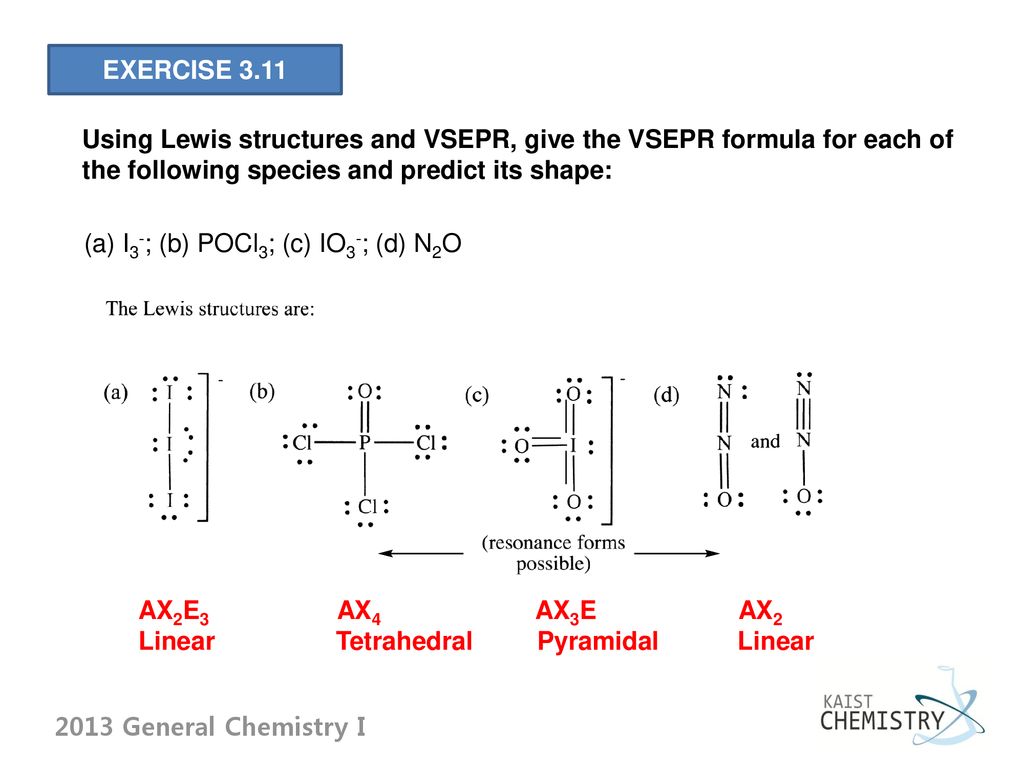
The Structure and Bonding of IO3- An example of the use of Lewis Structures and VSEPR Theory Lecturer: Dr. Andreas Lemmerer. - ppt video online download

SOLVED: In the IO3- ion, iodine is the central atom. Based on the Lewis structure of IO3- that obeys to the Octet rule, what is the formal charge on the iodine atom?

For the IO3-, a) Draw the electron dot diagram and structural formula b) Predict the shape c) Predict whether it is polar or non-polar, and justify your prediction. Indicate the positive and

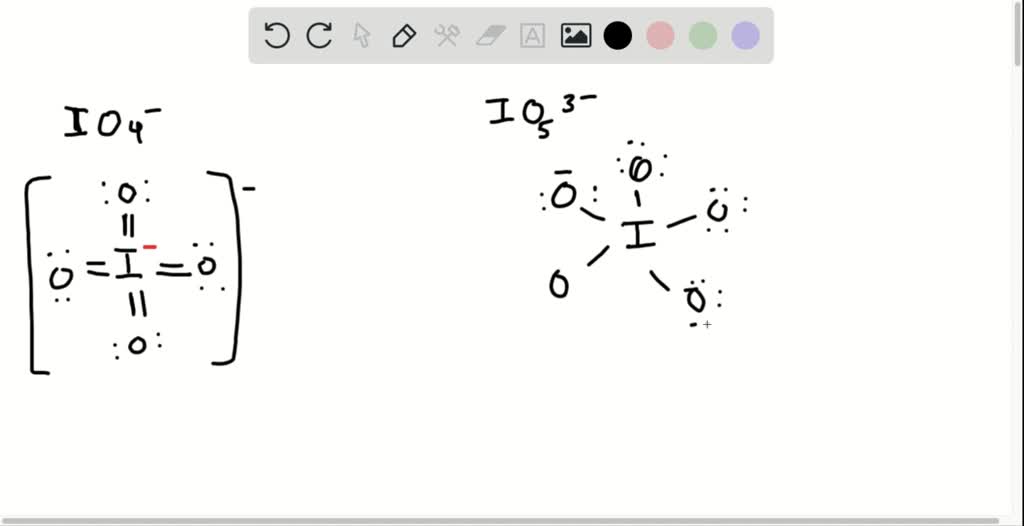
SOLVED: Draw the Lewis structure for BrO4-. What is the hybridization of the central atom (2p orbitals)? sp3 12. Determine the hybridization at each of the three atoms (C, O, E) from

Why the bond angle of XeO3 is 103 - Chemistry - Chemical Bonding and Molecular Structure - 12894693 | Meritnation.com
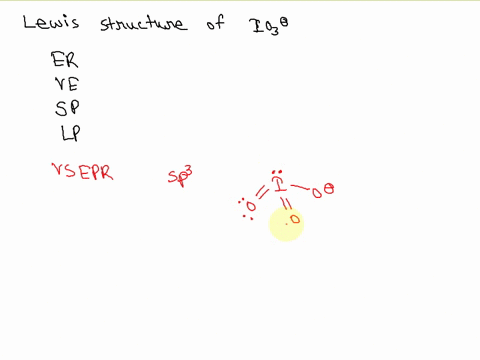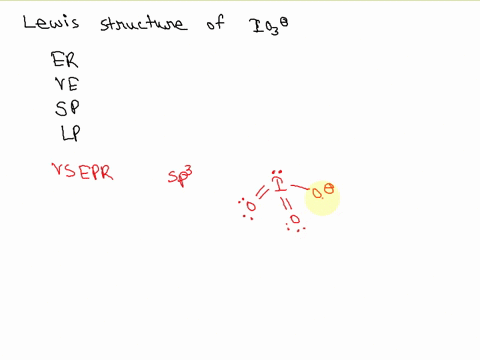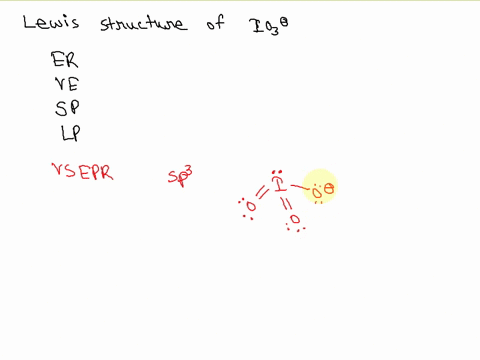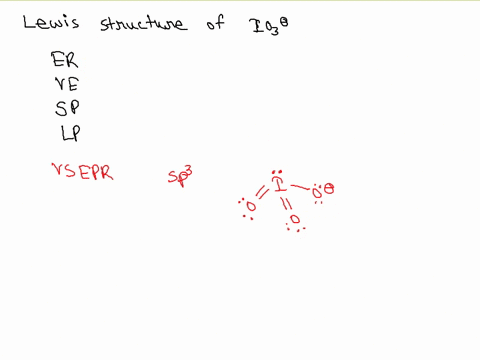








![hybridization process for charged molecule. example [IO3]- - YouTube hybridization process for charged molecule. example [IO3]- - YouTube](https://i.ytimg.com/vi/s39QIsfZZlI/maxresdefault.jpg)