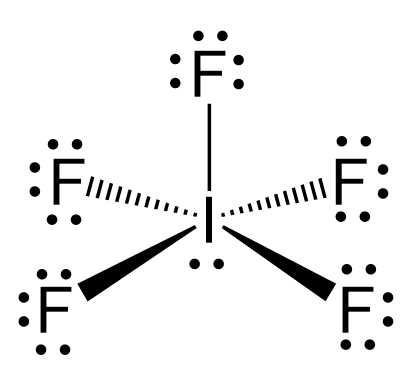
![Shape and hybridization of \\[{\\text{I}}{{\\text{F}}_{\\text{5}}}\\] respectively are ______.A ) trigonal bipyramidal, \\[s{p^3}d\\]B ) see-saw \\[s{p^3}d\\]C ) square pyramidal, \\[s{p^3}{d^2}\\]D ) pentagonal pyramidal \\[s{p^3}{d^3}\\] Shape and hybridization of \\[{\\text{I}}{{\\text{F}}_{\\text{5}}}\\] respectively are ______.A ) trigonal bipyramidal, \\[s{p^3}d\\]B ) see-saw \\[s{p^3}d\\]C ) square pyramidal, \\[s{p^3}{d^2}\\]D ) pentagonal pyramidal \\[s{p^3}{d^3}\\]](https://www.vedantu.com/question-sets/14baae8b-19c1-486a-aeeb-fe0e96903d743424253312437900136.png)
Shape and hybridization of \\[{\\text{I}}{{\\text{F}}_{\\text{5}}}\\] respectively are ______.A ) trigonal bipyramidal, \\[s{p^3}d\\]B ) see-saw \\[s{p^3}d\\]C ) square pyramidal, \\[s{p^3}{d^2}\\]D ) pentagonal pyramidal \\[s{p^3}{d^3}\\]

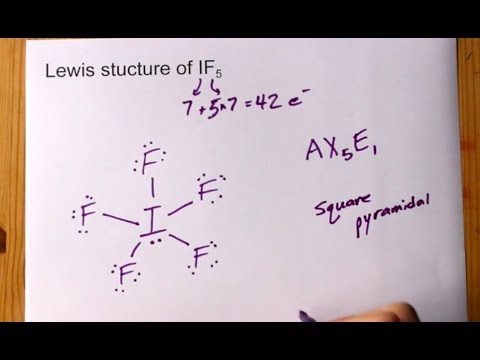
Draw the Lewis structure for IF5 and answer the following questions. a. How many valence electrons are there? b. What is the electron geometry? c. What is the molecular geometry? d. What
Shape and hybridisation of IF5 are ……… (a) Trigonal bipyramidal, sp^3d^2 (b) Trigonal bipyramidal, sp^3d - Sarthaks eConnect | Largest Online Education Community

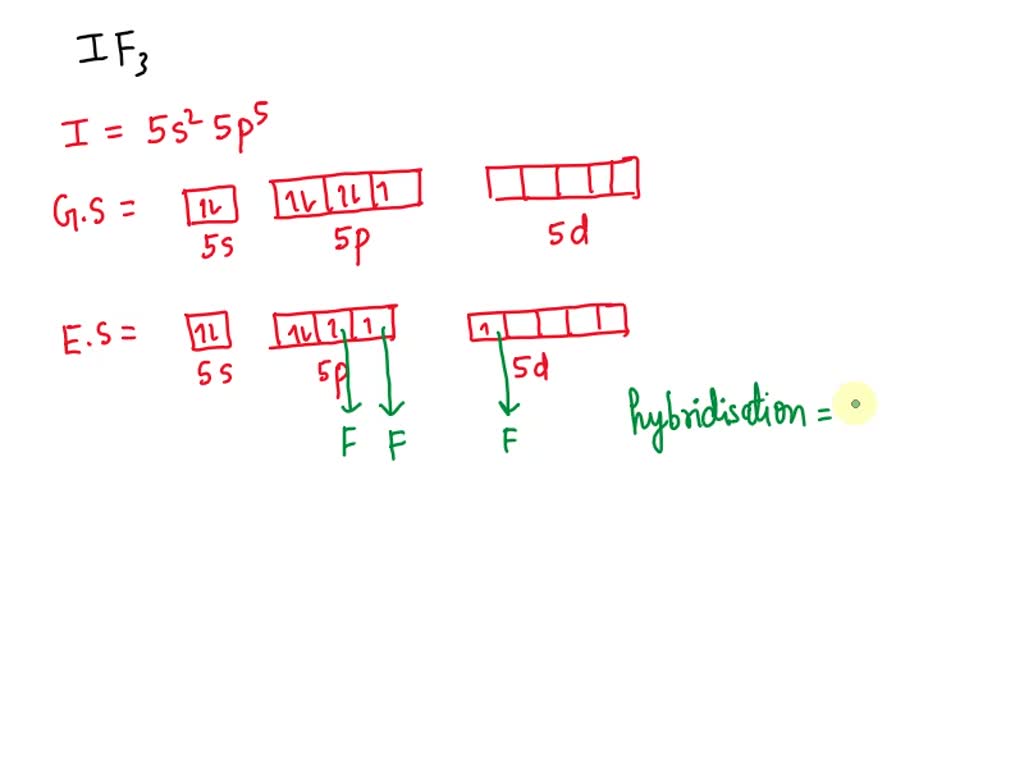
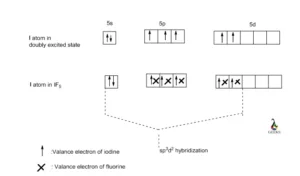
12. Which of the following compounds exhibit d 2sp 3 hybridization? (Select all that apply.) BrF5 ClF5 KrCl4 XeCl2 PCl5 13. Draw the Lewis structure for IF3 and answer the following question.



![ANSWERED] A. What is the hybridization of the central atom in IF5? - Kunduz ANSWERED] A. What is the hybridization of the central atom in IF5? - Kunduz](https://media.kunduz.com/media/sug-question/raw/52262186-1659250506.6599846.jpeg)