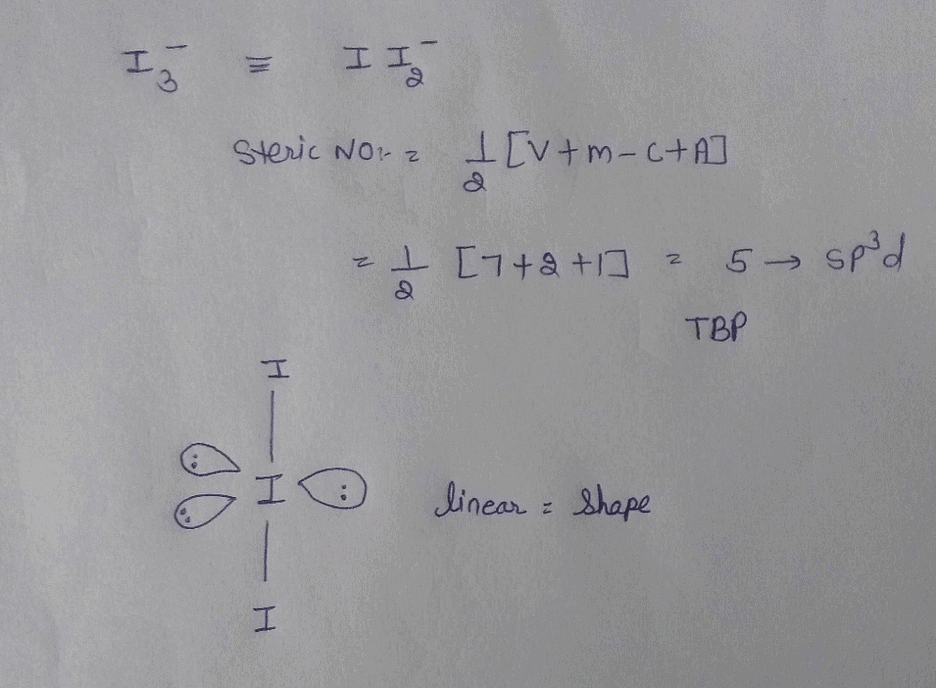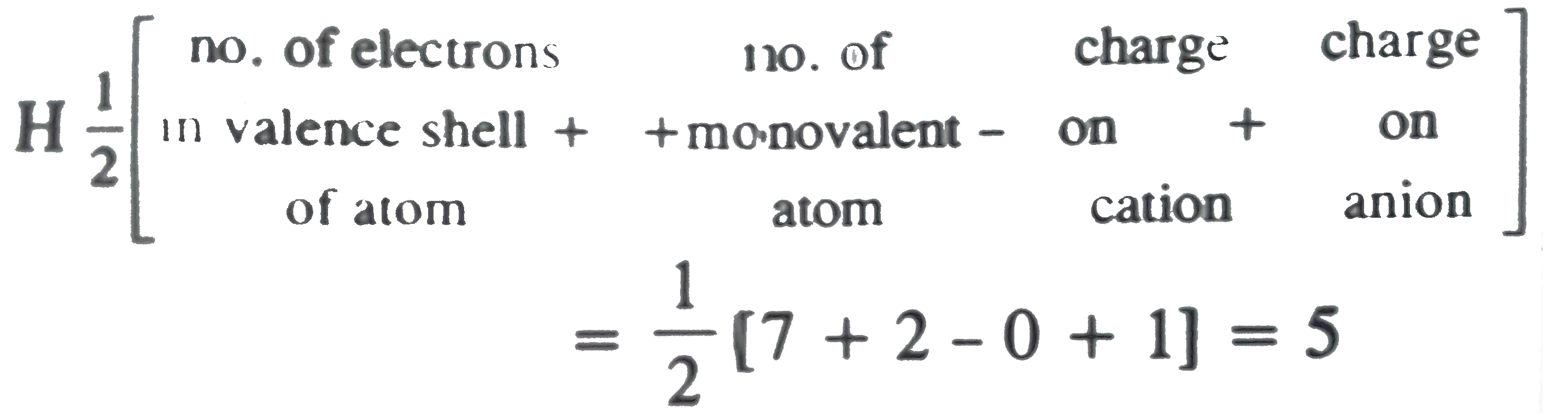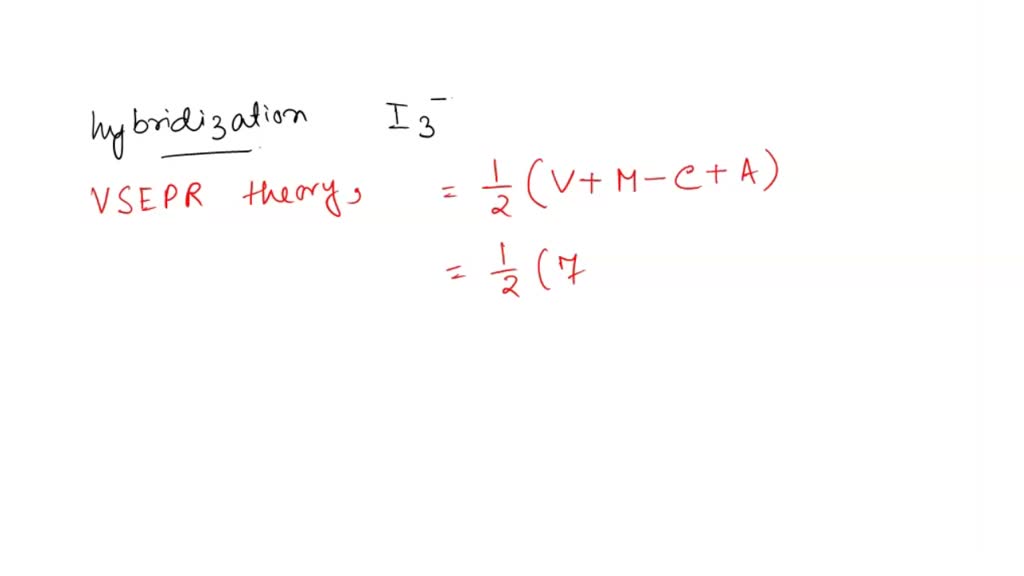Draw structure and state hybridization, geometry and shape of1) I3+ and 2) I3- - Chemistry - The p-Block Elements - 14318665 | Meritnation.com

Welcome to Chem Zipper.com......: What are the hybridixation and shape of I3- and I3+ PolyIodide ions!

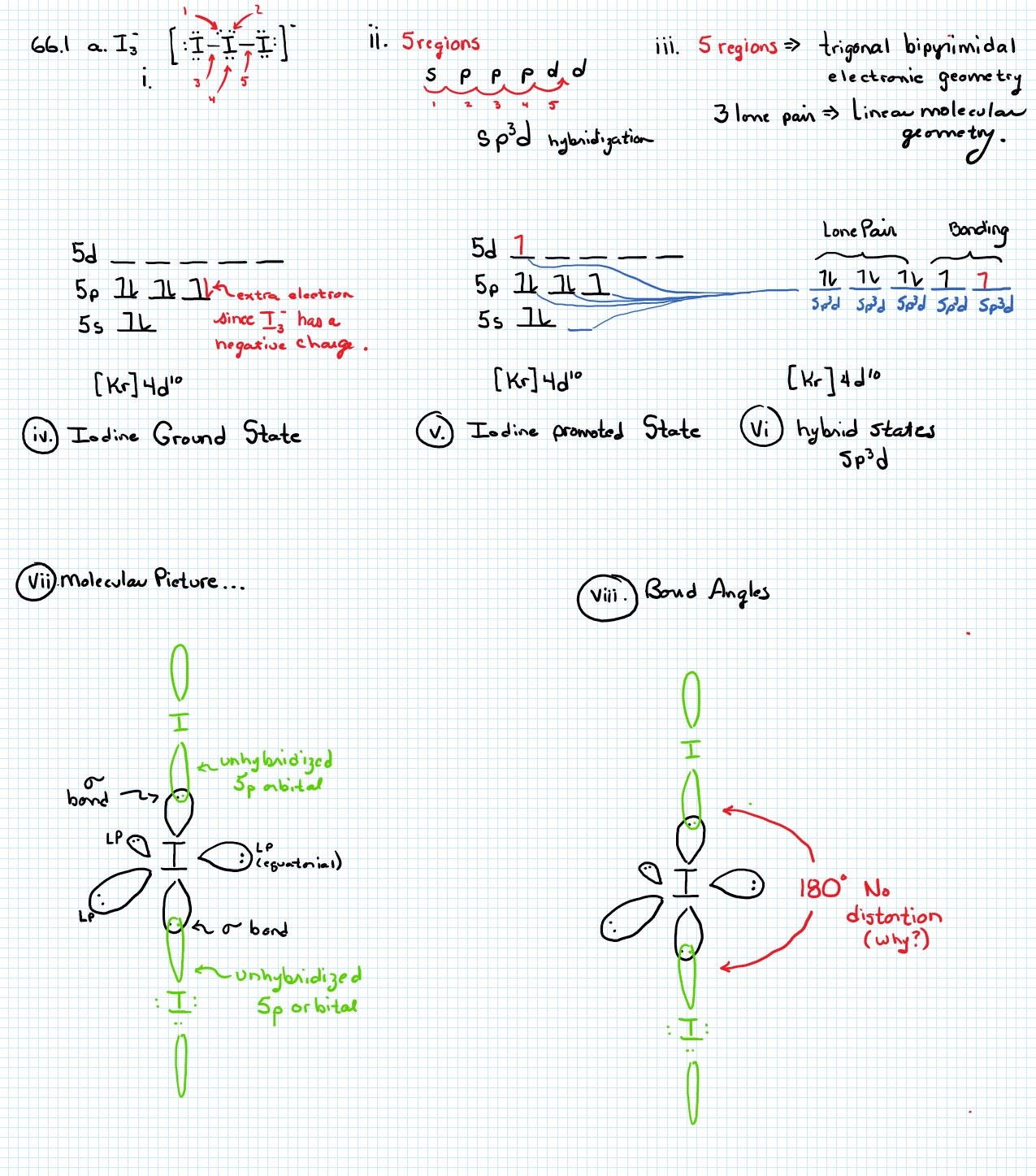
The hybridization states of the central atoms of the ions I3, ICI, and ICI, are respectively : (1) sp', dsp?, sp (2) spºd, spºd? and spºd (3) sp'd, spºd, dsp? (4) sp, sp, dsp?

The hybridization, lone pair of electrons and shape of {I}_3^- is:it has sp^3d hybridizationit has trigonal bipyramidal shapeit is linearit has 3 lone pair of electrons

Welcome to Chem Zipper.com......: Number of lone pair present at central atom of I3- ion and also give hybridisation and shape of ion?